I'm pregnant and a doctor: This is why I got the COVID-19 vaccine
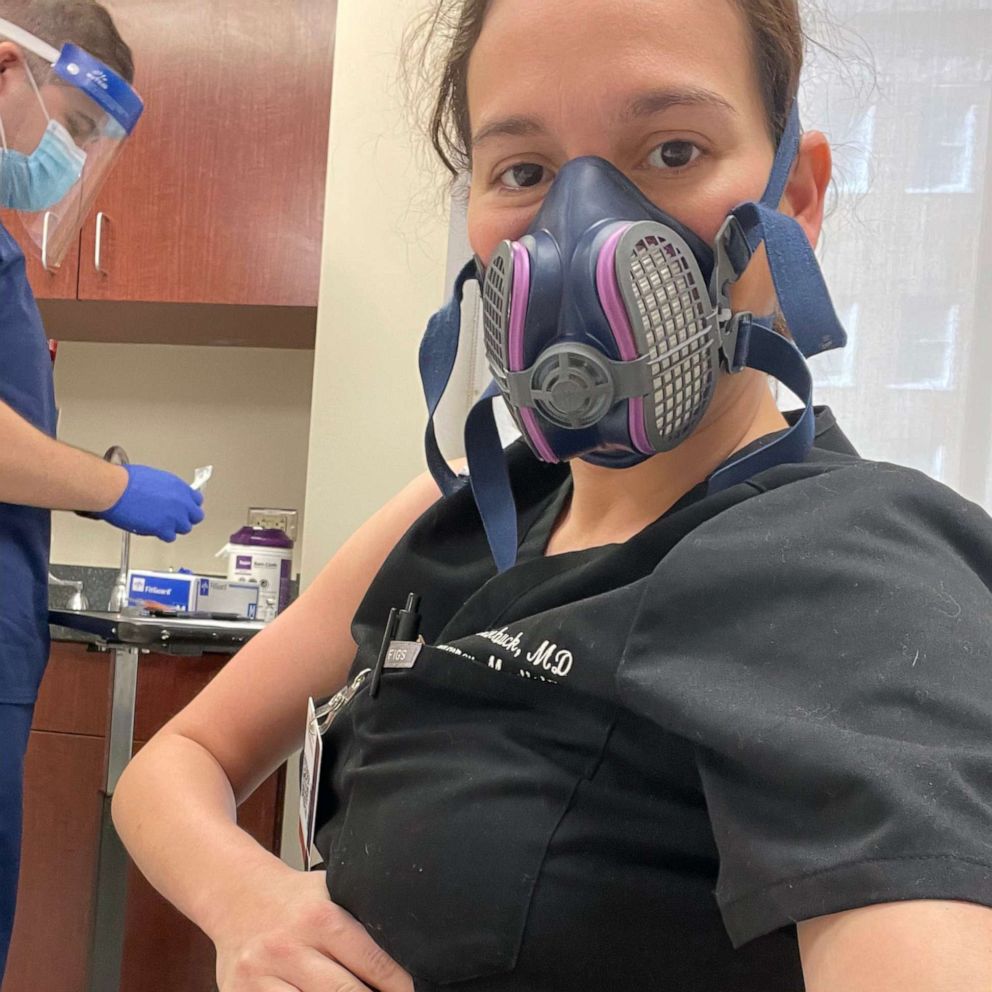
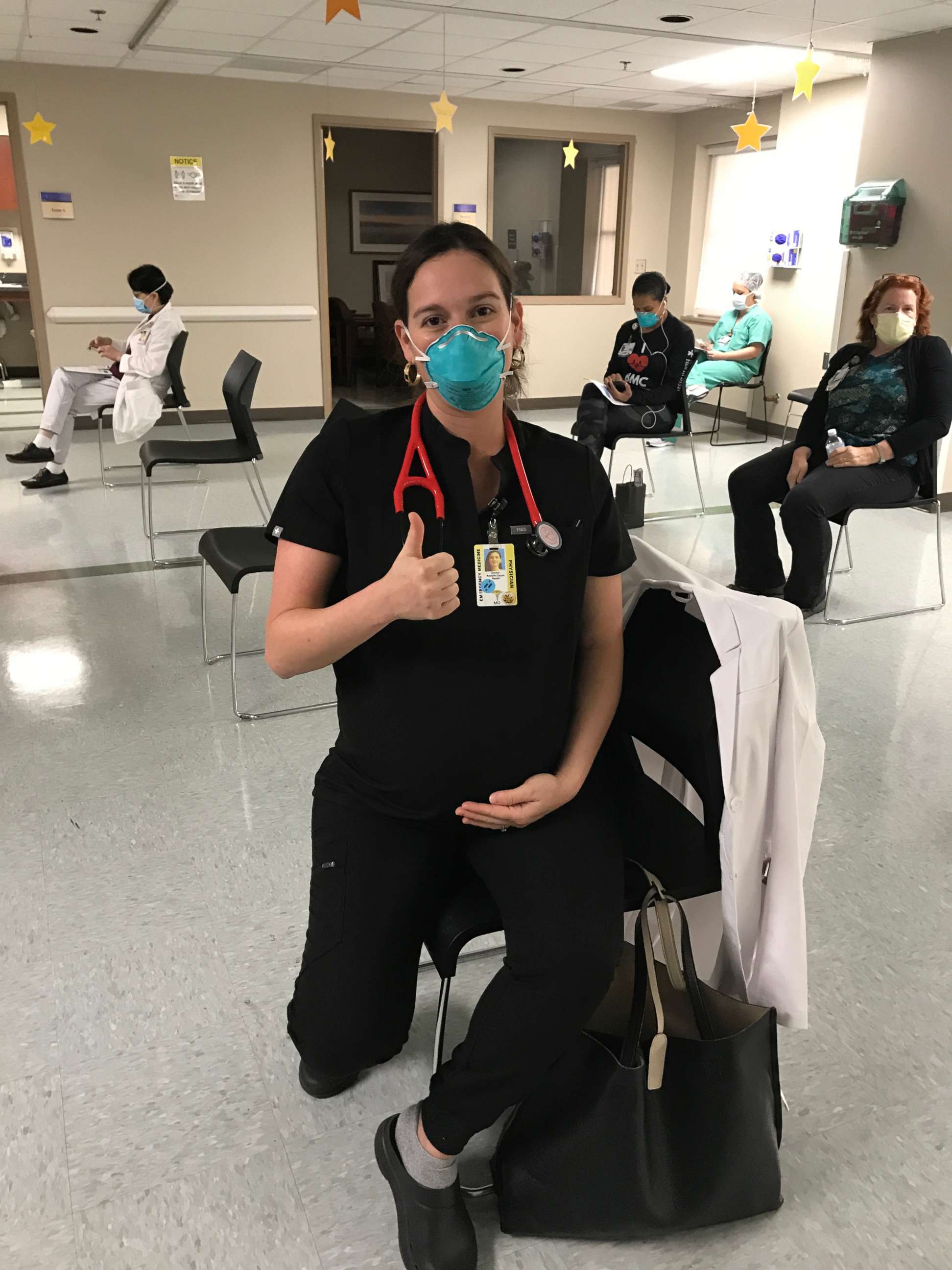
Dr. Nayeli Rodulfo-Zayas, an emergency medicine physician in San Antonio, Texas, was 35 weeks pregnant when she received her second dose of the COVID-19 vaccine earlier this month.
Rodulfo-Zayas, also the mother of a 2-year-old, is speaking out about her decision in hopes of inspiring other pregnant women to have important, educated conversations with their health care providers about getting the vaccine.
Pregnant women have not been actively included in the late-stage clinical trials for any COVID-19 vaccines, including the Pfizer-BioNTech and Moderna vaccines in the U.S.
Both the Pfizer and Moderna vaccines use mRNA technology, which uses pieces of viral genetic material as a way for the body to learn and develop defenses against future infection. If approved, they would be the first mRNA vaccines, which are theoretically safer during pregnancy because they do not contain a live virus.
Rodulfo-Zayas said knowing the science of mRNA technology -- which does not enter the nucleus of the cells and doesn’t alter the human DNA -- helped inform her decision to get vaccinated against COVID-19.
"That in itself is one of the reasons why I decided to take the vaccine, because I understand the science and I’m confident in the science, and also because I’m at high risk of catching the disease," she said. "I’m a Hispanic woman and I work around COVID patients constantly."
"I had a discussion with my obstetrician-gynecologist and we decided that the best thing for me was to get the vaccine," she added.
The decision to get the vaccine was also a very personal one for Rodulfo-Zayas, who lost her 57-year-old mom to the virus last year.

"She contracted COVID probably in early June and unfortunately she was part of that vulnerable population," said Rodulfo-Zayas, explaining that her mom had diabetes, high blood pressure and was on dialysis. "She was doing OK for about a week and then she got really sick and ended up in the ICU."
"In six more months, she could have gotten her vaccine," she added of the timing of her mom's death. "It’s a huge loss for me. I miss her every day, but I know she would have wanted me to do good and push through and continue doing the work I do and that’s why I’m doing this and I’m educating people."
Rodulfo-Zayas received her first dose of the vaccination in mid-December and her second dose on Jan. 7. She said she did have some minor side effects from the second shot, including chills and a low-grade fever that went away in less than 24 hours.
"I just want people to know that the [vaccine] side effects are minimal, that they do exist, they do make some people feel sick, but it’s temporary, and it really doesn’t compare at all what I’ve seen people go through when they get COVID and they are hospitalized," she said. "They pale in comparison to what people go through when they get COVID and need oxygen."
The U.S. Centers for Disease Control and Prevention (CDC) says that women who are pregnant and in the priority groups recommended to receive the COVID-19 vaccine "may choose to be vaccinated" during pregnancy and can make an informed decision by speaking with their health care provider to better understand the potential risks and benefits.
The American College of Obstetricians and Gynecologists (ACOG), a professional membership organization for OB-GYNs, issued a practice advisory last month to say that both pregnant and breastfeeding women who are in the priority groups for the vaccine should receive doses as well.
Rodulfo-Zayas said she urges other pregnant people to speak with their health care providers about whether they should receive the COVID-19 vaccine and to consider the risks of not getting vaccinated.
Data shows that pregnant people with COVID-19 are at an increased risk for "intensive care unit admission, invasive ventilation, extracorporeal membrane oxygenation, and death," compared to nonpregnant people, according to the CDC.
"I would just encourage people to get vaccinated, especially if you’re immunocompromised or you have chronic medical conditions because catching COVID has a lot of repercussions," said Rudolfo-Zayas. "COVID itself is really dangerous. The vaccine side effects, not so much."
What to know about pregnancy and the COVID-19 vaccine
The question of whether a pregnant person should receive a COVID-19 vaccine will eventually come down to a number of factors, including everything from the trimester, risk factors for COVID-19, ability to remain socially distanced in her lifestyle and occupation, guidance from federal and state officials and recommendations from one's own physicians, experts say.
Health experts will need to rely on continuously incoming data to make decisions around how safe the COVID-19 vaccines are during pregnancy.
Officials are doing the same for the general population, considering the speed at which the COVID-19 vaccines were developed, and people who are pregnant should not be "unnecessarily alarmed," Dr. Ruth Faden, the founder of the Johns Hopkins Berman Institute of Bioethics and a bioethicist who studies the ethics of pregnancy and vaccines, told "Good Morning America" last year.
"Vaccines are coming, and they're coming before we have all of the information that we would like to have to make recommendations for pregnant people," she said. "But this is the context in which decisions are being made and recommendations are being made for the general population, without all of the evidence we would like to have, which is why the studies will continue."
The Society for Maternal-Fetal Medicine (SMFM) released a statement in December encouraging expecting parents to "engage in shared decision-making" about the vaccine with their doctors.
"In general, SMFM strongly recommends that pregnant women have access to COVID-19 vaccines in all phases of future vaccine campaigns, and that she and her health care professional engage in shared decision-making regarding her receipt of the vaccine. ... mRNA vaccines, which are likely to be the first vaccines available, do not contain a live virus but rather induce humoral and cellular immune response through the use of viral mRNA," the society said in its statement. "Health care professionals should also counsel their patients that the theoretical risk of fetal harm from mRNA vaccines is very low."
Health experts say pregnant people with or without the vaccine need to continue to remain on high alert when it comes to COVID-19 by following safety protocols, including face mask wearing, social distancing and hand washing.
"What I tell my patients is the one thing I know for sure is that pregnant women can get COVID," Dr. Rashmi Rao, a maternal-fetal medicine specialist at UCLA Medical Center in Los Angeles, told "GMA" last year. "We’re not seeing huge rates of any adverse fetal outcomes yet, but, again, that’s still to be determined, because we have a long way to go with collecting information."
ABC News' Sony Salzman and Eric Strauss contributed to this report.